Introduction: Local treatment with intratumoral CpG (a toll-like receptor 9 agonist, SD-101) and low-dose radiation can elicit antitumor immune responses and global tumor reduction in patients with low-grade lymphoma (Frank, Cancer Discov, 2018). Ibrutinib compromises B-cell survival by inhibiting Bruton's tyrosine kinase, but also modulates T-cells by inhibiting interleukin-2-inducible T-cell kinase. In a mouse model of lymphoma, ibrutinib plus intratumoral CpG was curative of systemic disease, an effect that was T-cell dependent (Sagiv-Barfi, Blood, 2015). Thus, we initiated a phase I/II clinical trial combining oral ibrutinib, intratumoral CpG and local low-dose radiation in adults with recurrent low-grade lymphoma (NCT02927964).
Methods: Enrolled patients received intratumoral injections of CpG (SD-101, 3mg) weekly for 5 doses, starting on the second day of a 2-day course of local radiation (4Gy total) to the same site. Daily oral ibrutinib (560mg) began on day 9. Treatment-emergent adverse events (AEs), ibrutinib dose modifications and adherence were recorded at every visit. Revised Lugano criteria (Cheson et al., JCO, 2014) were used to assess response to therapy, based on CT scans at 3, 6, 12, 18, and 24 months. Fine needle aspirates (FNAs) were obtained from CpG-injected and non-injected nodal tumor sites pre- and post-treatment and analyzed by flow cytometry and single-cell RNA sequencing (scRNAseq). When available, viably preserved tumor and peripheral blood cells were used for in vitro immune response assays.
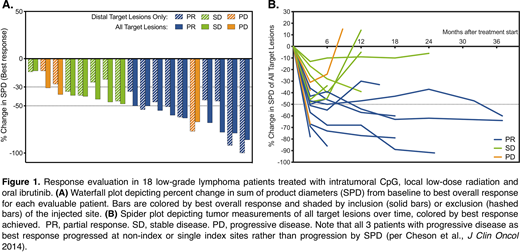
Results: As of July 16, 2020, 18 patients had been treated, with a median follow-up of 12 months. Ten were male and 8 were female. All but one had a diagnosis of follicular lymphoma; one patient had marginal zone lymphoma. All were previously treated with an average of 2 prior lines of therapy. AEs were consistent with known effects of ibrutinib (including diarrhea and rash) and of CpG (including fever and flu-like reaction) with no unexpected AEs to suggest synergistic toxicity. There were no grade 4 or 5 events. AEs led to ibrutinib dose reduction or discontinuation in 2 patients and dose interruption in 6 patients. At the time of analysis, 9 of 18 evaluable patients had achieved a partial response (50% ORR) and 12 of 18 patients experienced at least a 30% reduction in the distant uninjected lesions (Figure 1A). Most responses have been maintained for at least 6 months, many longer (Figure 1B). Flow cytometry revealed decreased T follicular helper cells and increased CD4 and/or CD8 effector T-cells, CD137+ activated T-cells, and NK cells in CpG-injected tumors. Abscopal immune effects in distant non-injected lesions included an increase in Granzyme B+ CD8 T-cells, most prominent after the addition of ibrutinib. scRNAseq data showed significant transcriptional shifts in tumor cells and in tumor-infiltrating T-cells, including signatures of interferon response and T cell activation and cytotoxicity. Finally, in vitro assays showed tumor-specific immune responses in peripheral blood T-cells of all 6 evaluable patients tested thus far.
Conclusion: Early data suggest that the combination of oral ibrutinib, intratumoral CpG, and local low-dose radiation is safe and can generate systemic antitumor immune responses and systemic tumor shrinkage in low-grade lymphoma.
Khodadoust:Seattle Genetics: Consultancy; Kyowa Kirin: Consultancy. Levy:Quadriga: Membership on an entity's Board of Directors or advisory committees; Beigene: Membership on an entity's Board of Directors or advisory committees; GigaGen: Membership on an entity's Board of Directors or advisory committees; Teneobio: Membership on an entity's Board of Directors or advisory committees; Sutro: Membership on an entity's Board of Directors or advisory committees; Checkmate: Membership on an entity's Board of Directors or advisory committees; Nurix: Membership on an entity's Board of Directors or advisory committees; Dragonfly: Membership on an entity's Board of Directors or advisory committees; Abpro: Membership on an entity's Board of Directors or advisory committees; Apexigen: Membership on an entity's Board of Directors or advisory committees; Spotlight: Membership on an entity's Board of Directors or advisory committees; 47 Inc.: Membership on an entity's Board of Directors or advisory committees; XCella: Membership on an entity's Board of Directors or advisory committees; Immunocore: Membership on an entity's Board of Directors or advisory committees; Walking Fish: Membership on an entity's Board of Directors or advisory committees; Viracta: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.